Electricity-driven nitrogen insertion enables sustainable heterocycle synthesis
December 19, 2025Researchers from the National University of Singapore (NUS) have developed an electrochemical reaction manifold that promotes efficient nitrogen atom insertion into saturated carbocycles to access either functionalised quinolines or N-alkylated saturated N heterocycles, both of which are privileged scaffolds in synthetic chemistry and pharmaceutical science.
The research team was led by Associate Professor KOH Ming Joo and Professor ZHAO Yu from the NUS Department of Chemistry. The research breakthrough was published in the scientific journal Nature Synthesis.
N heterocycles have garnered significant attention from academia and industry as building blocks for drug development. Therefore, developing practical and efficient procedures to access these motifs remains a central pursuit in synthetic chemistry. Among various N-heterocycle synthetic strategies, skeletal editing of readily available cyclic compounds through nitrogen atom insertion represents an unconventional and highly desirable approach. However, direct nitrogen atom insertion into saturated, unstrained carbocycles via C–C single-bond cleavage remains rare due to the high thermodynamic and kinetic stability of these σ bonds. Existing methods of nitrogen atom insertion into cyclic hydrocarbons rely on the stoichiometric use of strong oxidising agents and suffer from limited functional group compatibility.
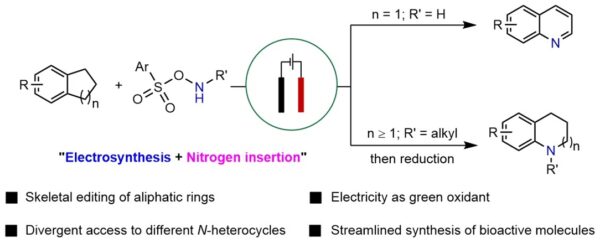
Divergent electrochemical nitrogen atom insertion into saturated carbocycles to access diverse valuable N heterocycles. [Credit: Nature Synthesis]
Assoc Prof Koh said, “We wanted to design a greener and controllable way of transforming saturated carbocycles into different classes of N heterocycles. This would greatly facilitate the sustainable synthesis of valuable N-heterocyclic bioactive compounds for applications such as drug discovery.”
The researchers devised a tunable electro-oxidative strategy for efficient nitrogen atom insertion into saturated carbocycles, enabling divergent access to either substituted quinolines or N-alkylated N heterocycles under ambient conditions. From a sustainability and environmental standpoint, the use of electricity to promote this redox transformation helps to reduce the generation of chemical waste. The method exhibits broad functional group tolerance, including oxidation-sensitive moieties, underscoring its adaptability and generality. The synthetic utility of this protocol is further demonstrated through the streamlined synthesis of two potential ion-channel antagonist candidates, highlighting its value for constructing bioactive N-heterocyclic frameworks.
“By strategically modulating the oxidation or reduction of the key cyclic imine intermediate, a broad array of substituted quinolines and N-alkylated N heterocycles bearing synthetically valuable functional groups can be efficiently accessed. We believe this methodology offers a versatile and robust platform for the modification and synthesis of bioactive scaffolds,” added Assoc Prof Koh.
Studies are ongoing to employ the methodology to synthesize other types of heterocyclic bioactive compounds.
Reference
Sun G; Wang X; Hu R; Rao W; Zhao Y*; Koh MJ*, “Divergent synthesis of N heterocycles from carbocycles enabled by electrochemical nitrogen atom insertion” Nature Synthesis DOI: 10.1038/s44160-025-00945-x Published: 2025.


